Signing of data is a feature of the RF500 Wireless Monitoring System, required for compliance to the electronic signature section of 21 CFR Part 11.
From the perspective of 21 CFR Part 11 the definition of an electronic signature is as follows:
12.2.2 Electronic Signature
A computer data compilation, of any symbol or series of symbols, executed, adopted or authorised by an individual to be a legally binding equivalent of the individual’s handwritten signature.
Signed data as far as the FDA is concerned means that the signer accepts that the data is true and good, and that he is taking legal responsibility for it. It is no different to printing off the page and signing it with a pen. It can be deemed as legally binding as shown above.
As far as RF500 is concerned, each historic Period of Data can be individually signed. A Period cannot be signed if it is still live and capable of storing extra data.
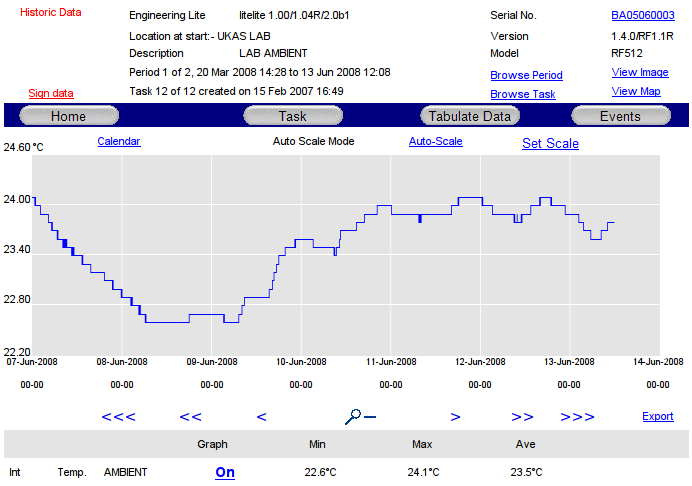
Find Data to Sign
Use the Browse Task and Browse Period links on the Transmitter Graph page, Transmitter Tabular Data page or Transmitter Events page, to find the historic Period to be signed.
In order to sign a live Period, that Period must be closed using either Start/Stop Logging or by Tasking the Transmitter. Only once it has become historic it is possible to sign the data.
To sign data, click the Sign Data link and the following page will be displayed:
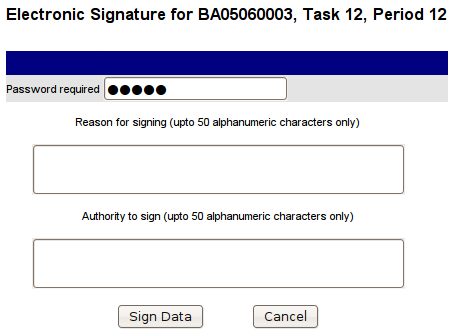
Finding Data to Sign
To sign data, enter the reason for signing the data and your authority to sign, enter your password and click the Sign Data button. The data is then digitally signed and the text entered is displayed on the Transmitter Graph page, Transmitter Tabular Data page and Transmitter Events page.
Each action of Data Signing is recorded in the Audit Trail.
