Wireless Temperature Monitoring Solutions
Vaccine Monitoring
All of our product ranges offer a solutions for ultra-low temperature storage, allowing you to monitor down to at least -328°F, making our solutions flexible enough to cope with whatever your vaccine requirement might be.
Comark is committed to supporting healthcare industries across the globe in the wake of the COVID-19 pandemic, to expand their capabilities and use our expertise and knowledge to provide dependable, precise solutions to critical temperature measurement issues.
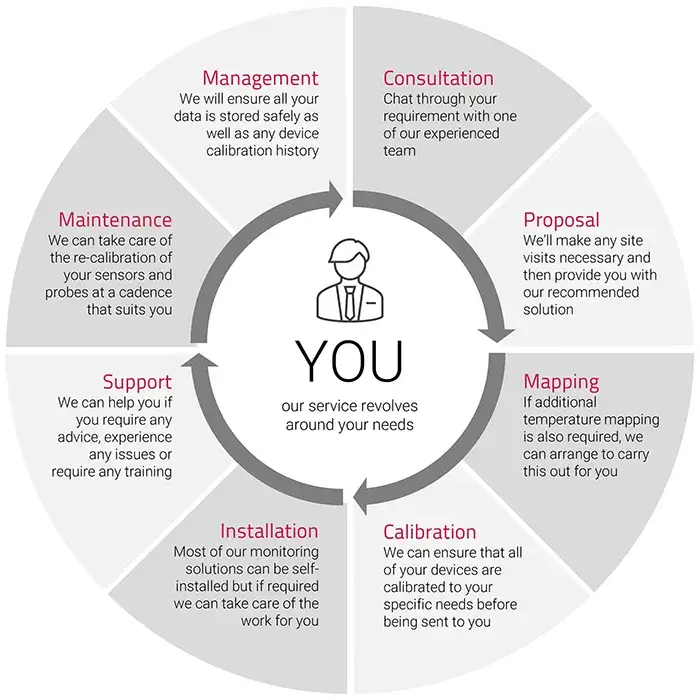
The Diligence 600 WiFi Monitoring System is our most advanced and most powerful static monitoring system yet. It provides a highly accurate, continuous temperature monitoring and analysis solution that utilizes your organization’s existing WiFi (Wireless) network.